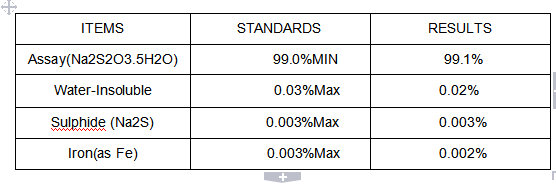
SOLVED: The compound sodium thiosulfate pentahydrate, Na2S2O3·5H2O, is important commercially to the photography business as "hypo", because it has the ability to dissolve unreacted silver salts from photographic film during development. Sodium

Uncaking cristaux incolores de qualité industrielle Na2S2O3 5H2O du thiosulfate de sodium - Chine Le thiosulfate de sodium anhydre, hyposulfite de sodium
What mass of Na2S2O3.5H2O is needed to make 500 cm^3 of 0.200 N solution for the reaction : - Sarthaks eConnect | Largest Online Education Community
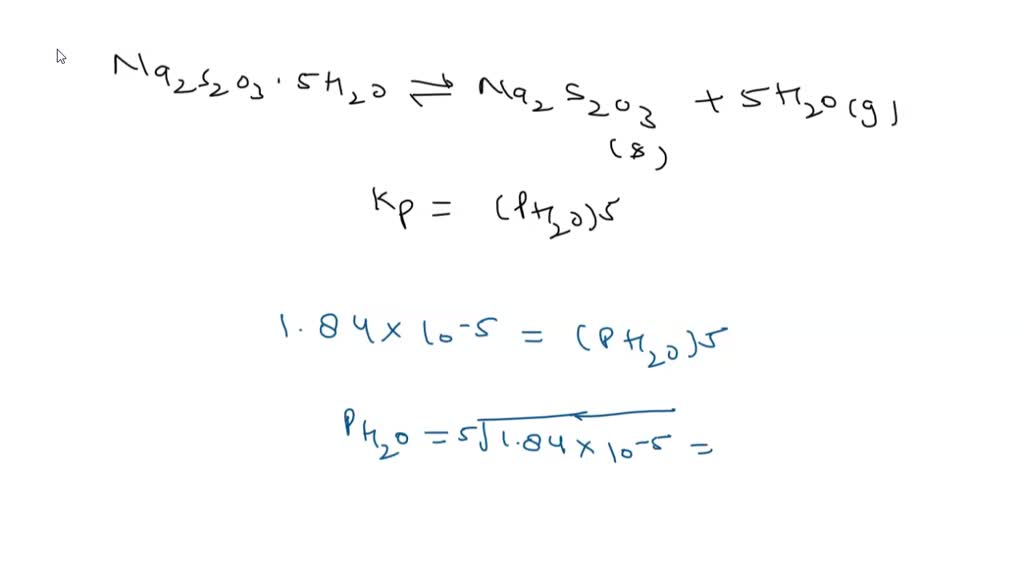
SOLVED: Sodium thiosulfate pentahydrate (FM 248.18 g ∙ mol-1) loses water when it is heated in an oven: Na2S2O3∙5H2O(s) ⇌ Na2S2O3(s) + 5H2O(g). ΔH° and ΔS° for this reaction at 25°C are

10102-17-7, 248.19, Sodium Thiosulfate, Pentahydrate, Crystal, Reagent, ACS - 6NPD7|S1500-500GM - Grainger