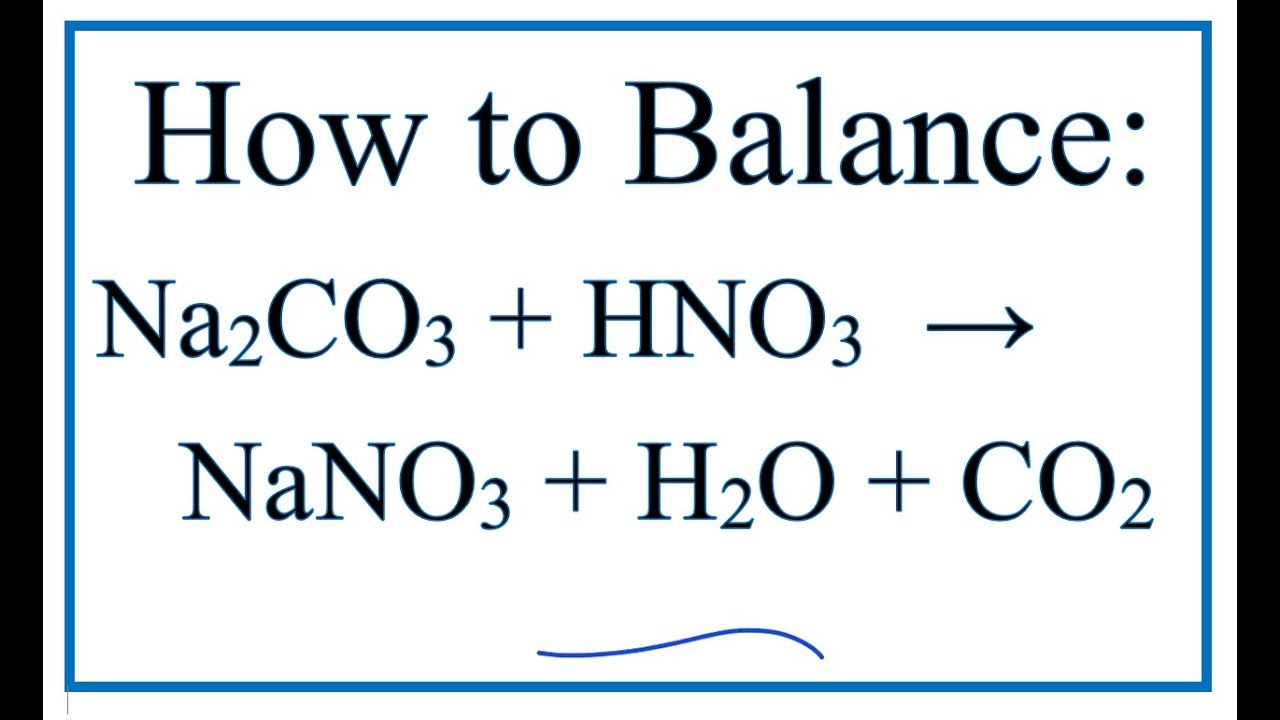
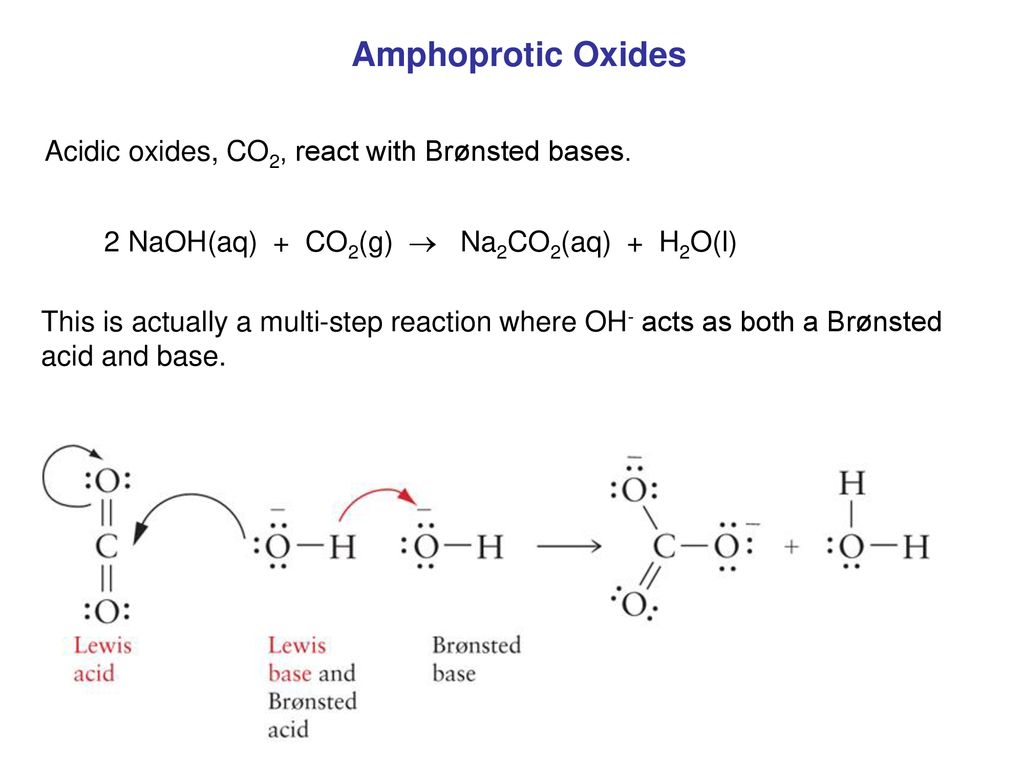
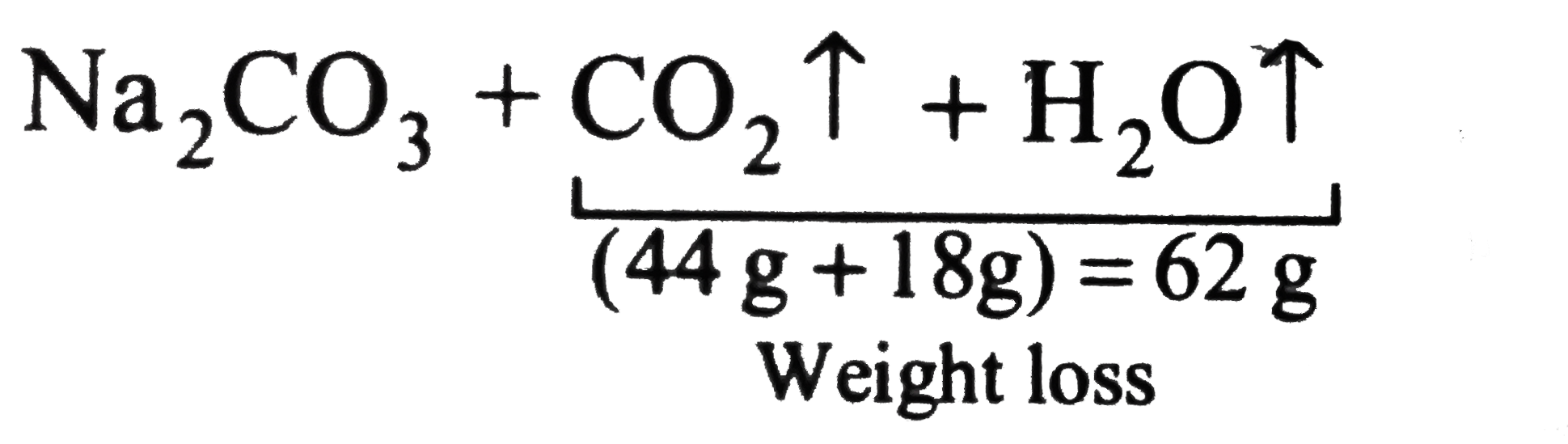Ответы Mail.ru: Химия 8 класс! NaOH+CO=Na2CO2+H2O правильная ли это реакция и еще как мне получить Na2CO2?

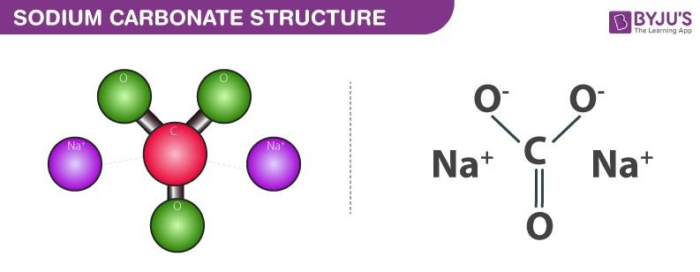
Sodium Carbonate (Na2CO3) Reaction: What does Sodium Carbonate do in a Reaction? - Aakash BYJU'S Blog

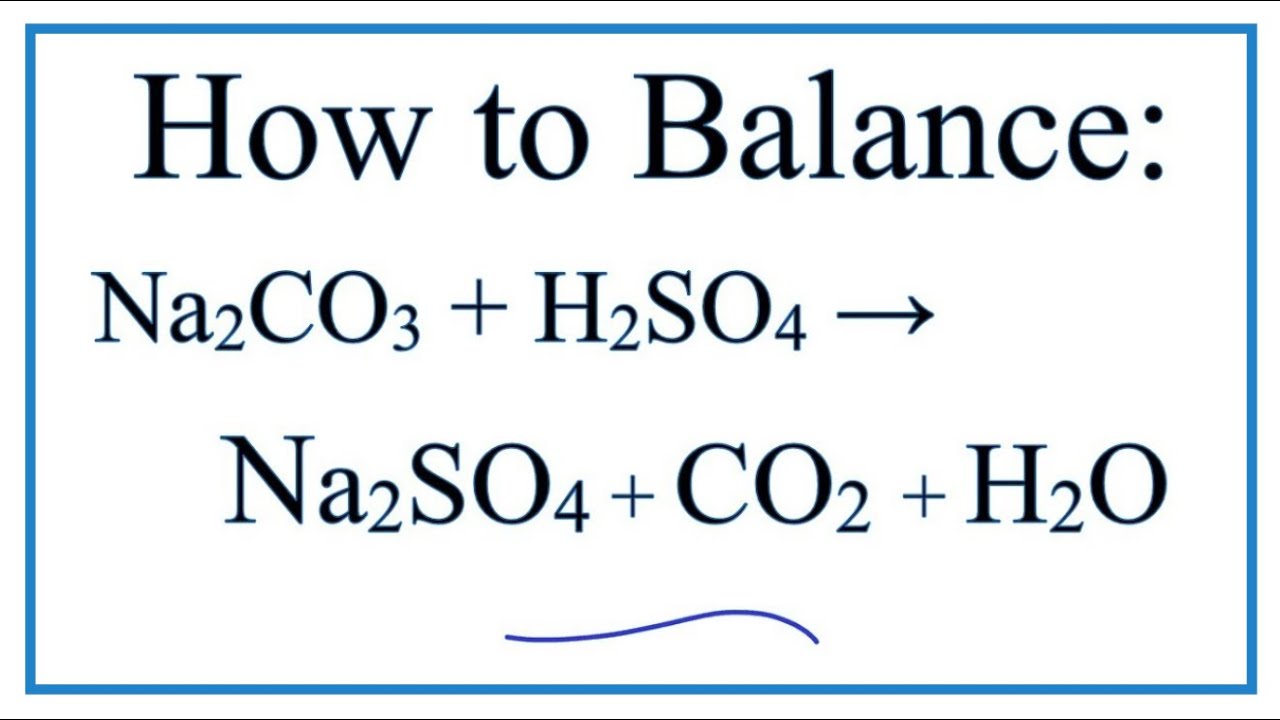
Na2CO3+HCl=NaCl+CO2+H2O Balanced Equation||Sodium carbonate+Hydrochloric acid=Sodium chloride+Carbon - YouTube

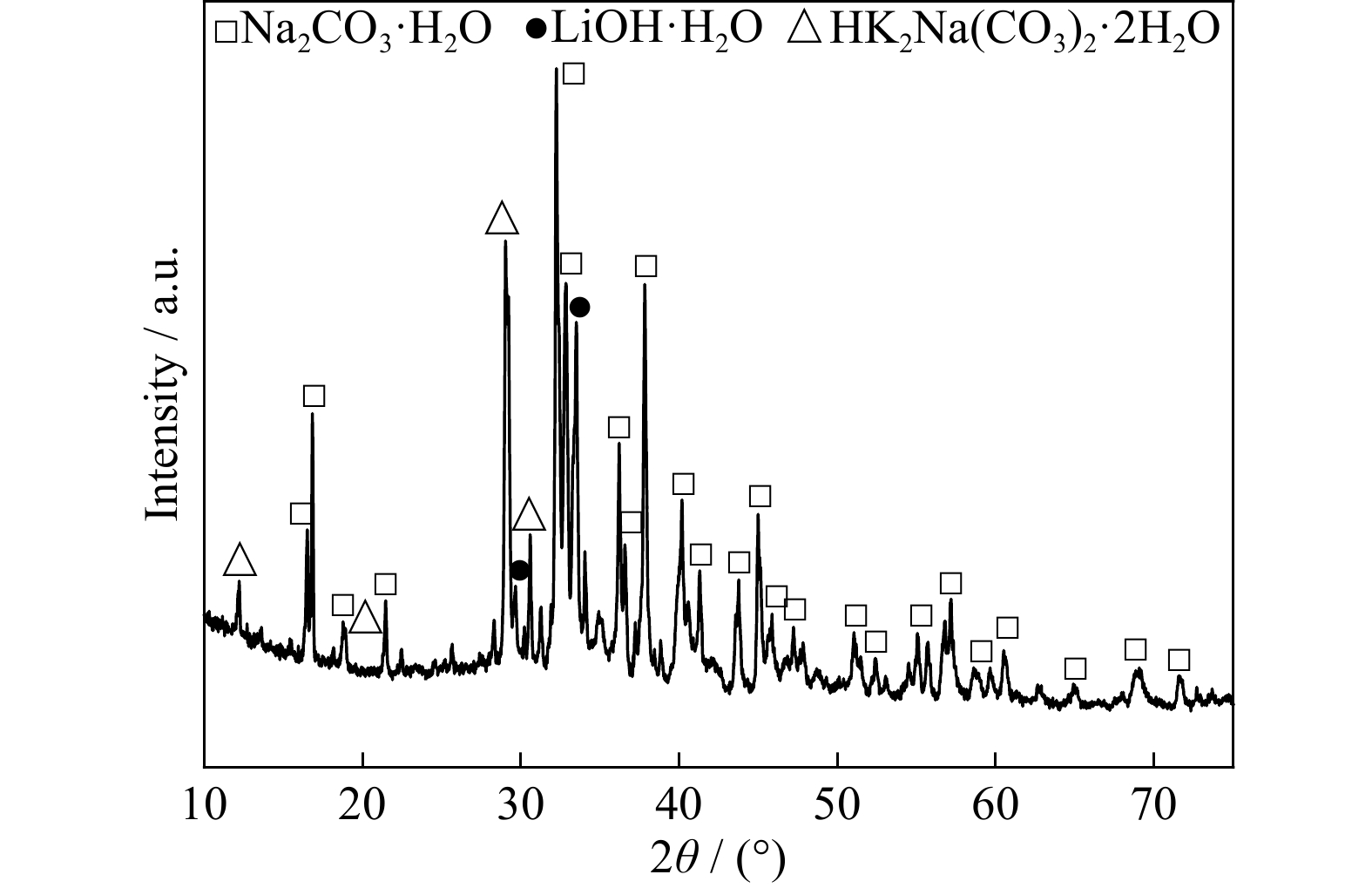
Membrane Crystallization of Sodium Carbonate for Carbon Dioxide Recovery: Effect of Impurities on the Crystal Morphology | Crystal Growth & Design
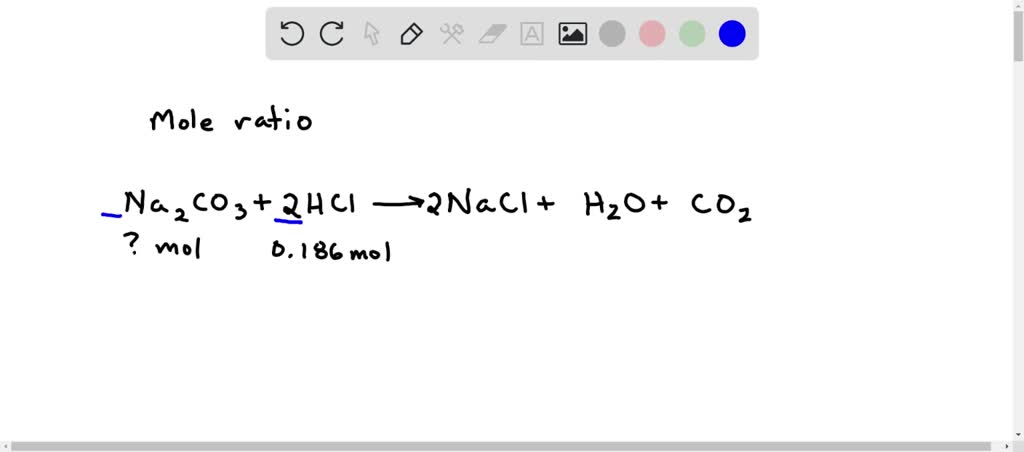
SOLVED: calculate the number of moles of sodium carbonate (Na2CO2) needed to neutralize 0,186 mole of HCI. chemical reaction : Na2CO3 + HCI —> NaCl + H20 + CO2

28. State as to why: (a) Aqueous solution of Na,co, is alkaline. (b) BaO is soluble but Baso, is insoluble in water. (1) Draw structure of BeClz (vapour). (m) Complete the following:

Washing soda has the formula Na_(2) CO_(2).10H_(2)O. What is mass of anhydrous sodium carbonate ... - YouTube

Na2CO3+HCl=NaCl+CO2+H2O Balanced Equation||Sodium carbonate+Hydrochloric acid=Sodium chloride+Carbon - YouTube

Phase Diagrams of Na2CO3–CO(NH2)2–H2O2–H2O System at 0 °C and 25 °C and the Production of Urea Peroxide and Sodium Percarbonate | Journal of Chemical & Engineering Data

A Strategy for Amide to β-Oxo Ester Transformation via N-Alkenoxypyridinium Salts as the Activator and H2O as the Nucleophile | Organic Letters