BFC BORAX LR - 500gm, DI-SODIUM TETRABORATE (Na2B4O7.10H2O) CAS No.1303-96-4 : Amazon.in: Industrial & Scientific
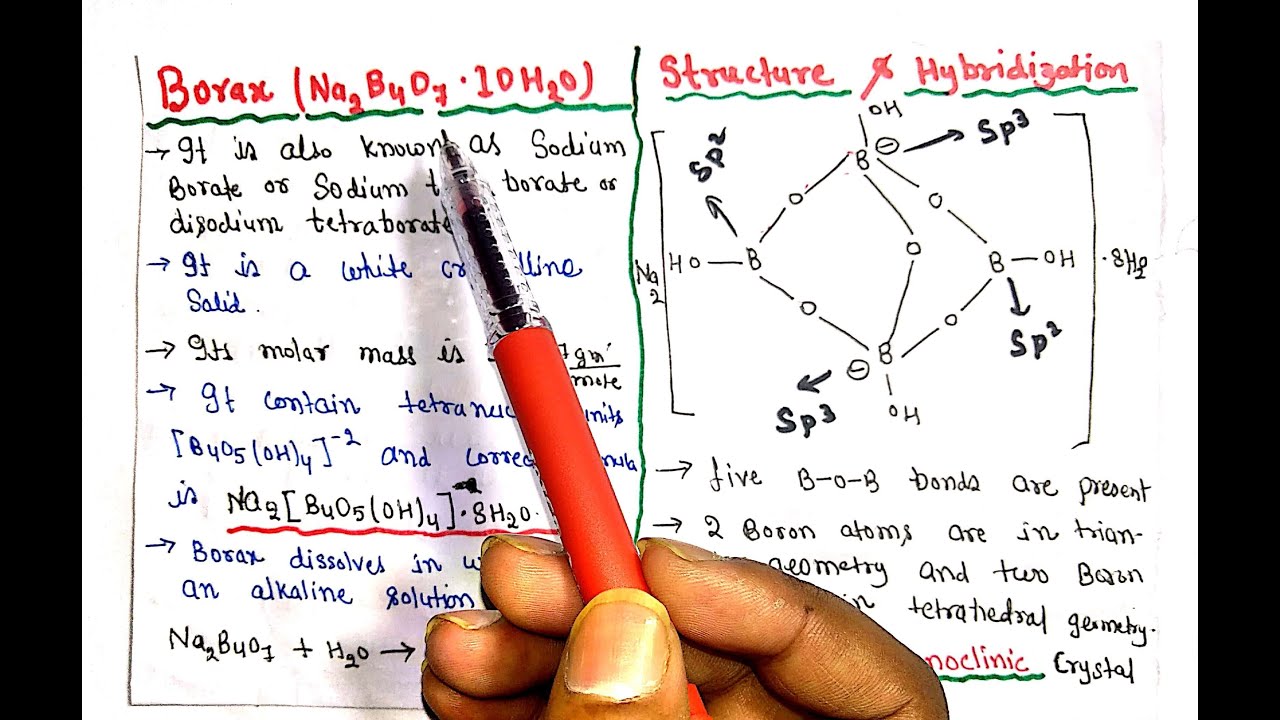
Borax (Na2B4O7•10H2O) || Properties || Structure & Hybridization || Preparation & Heating effect - YouTube

ขจัดเศษตัดเกรดทางเทคนิคให้น้ำ Na2b4o7.H2O 99.9 % - ชาวจีน น้ำสีทองสีเขียวแก้วสีทอง 99.5 สีทองเส้นร้อยละ 99.5 สีโซเดียมทรโบโรเสท โซเดียมเททโบโรเนน้ำเติมน้ำใสน้ำออกเทรไฮดะอันน้ำสูงชาน้ำใสสะอาดน้ำใสน้ำปราศจากน้ำน้ำน้ำน้ำน้ำน้ำน้ำน้ำไฮดรอสตีลเฟอร์ตีรัส ...

Eutectic Temperature, Density, and Solubility of H3BO3–H2O, Na2B4O7–H2O, and NaBO2–H2O Binary Systems | Journal of Chemical & Engineering Data
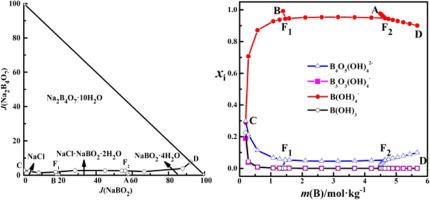
Solubility determination and thermodynamic modelling of solid−liquid equilibria in the (NaCl + NaBO2 + Na2B4O7 + H2O) system at 298.15 K,The Journal of Chemical Thermodynamics - X-MOL
Calculate the % of boron (B) in borax Na2 B4 O7 . 10H2O. (H = 1, B = 11, O = 16, Na = 23) - Sarthaks eConnect | Largest Online Education Community

Borax has the formula Na_{2}B_{4}O_{7}. 10H_{2}O. It is a strong base in aqueous solution because OH^{-} ions are produced by reaction with water.(B_{4}O_{7}^{2-}+7H_{2}O rightarrow 4H_{3}BO_{3}+2OH^{-}).How many grams of borax is necessary to








![Alchemy Alphabet: BORAX, also: Tinkal. Sodium... - Stock Illustration [76297336] - PIXTA Alchemy Alphabet: BORAX, also: Tinkal. Sodium... - Stock Illustration [76297336] - PIXTA](https://t.pimg.jp/076/297/336/1/76297336.jpg)