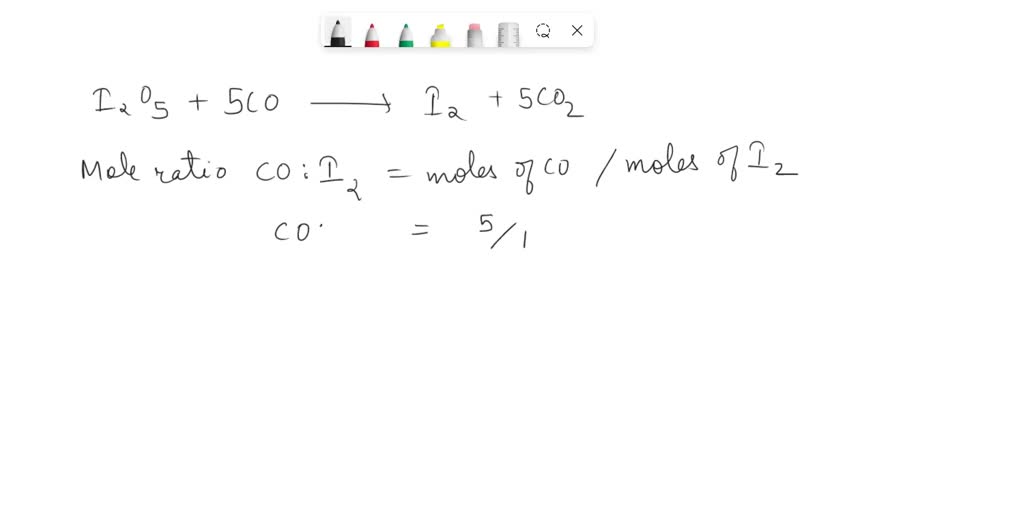
SOLVED: 1. I2O5 + 5CO —-> I2 + 5CO2 a) What is the mole to mole ratio of CO and I2? b) What is the mole to mole ratio of I2O5 and

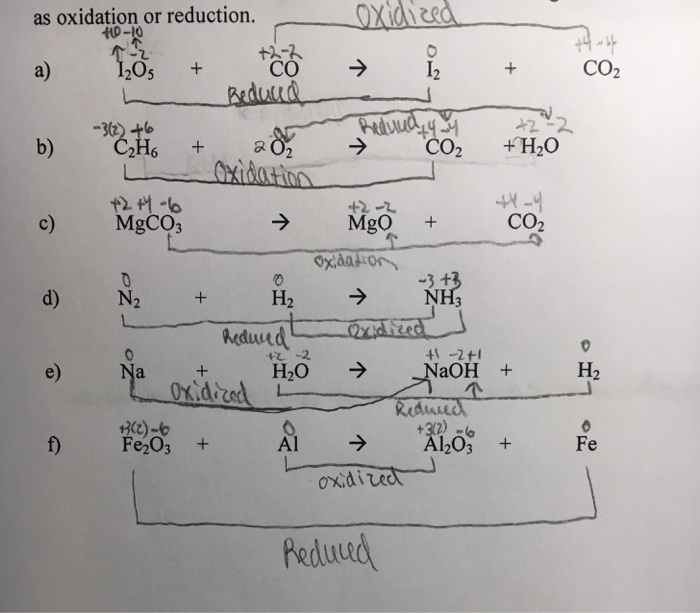
SOLVED: In each of the following balanced oxidation-reduction equations, identify those elements that undergo changes in oxidation number and indicate the magnitude of the change in each case. I2O5 (g) + 5

I2O5-Mediated 1,5-Cyclization of Aryldiynes with H2O: A Way To Access 3-Acyl-1-indenone Derivatives - ScienceDirect
![I2O5-Mediated Iodocyclization Cascade of N-(1-Arylallyl)pyridine-2-amines with Concomitant C═C Bond Cleavage: A Synthesis of 3-Iodoimidazo[1,2-a]pyridines | The Journal of Organic Chemistry I2O5-Mediated Iodocyclization Cascade of N-(1-Arylallyl)pyridine-2-amines with Concomitant C═C Bond Cleavage: A Synthesis of 3-Iodoimidazo[1,2-a]pyridines | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.9b00765/asset/images/medium/jo-2019-007654_0002.gif)
I2O5-Mediated Iodocyclization Cascade of N-(1-Arylallyl)pyridine-2-amines with Concomitant C═C Bond Cleavage: A Synthesis of 3-Iodoimidazo[1,2-a]pyridines | The Journal of Organic Chemistry