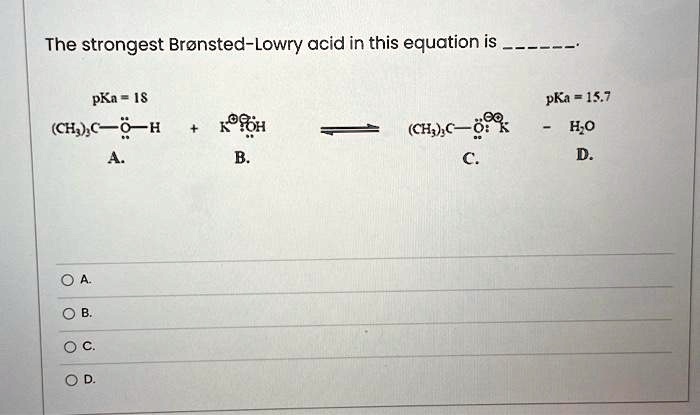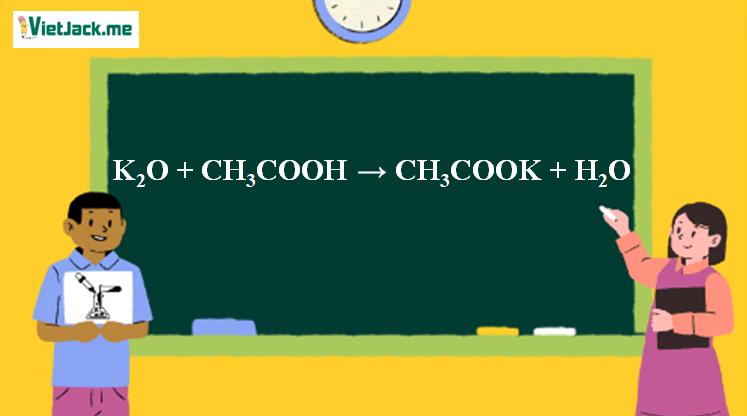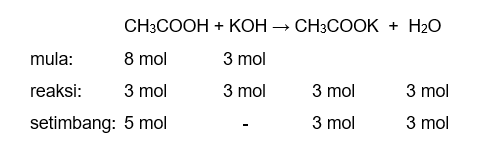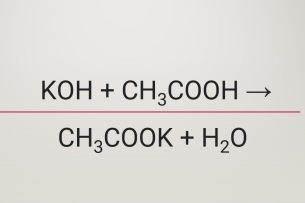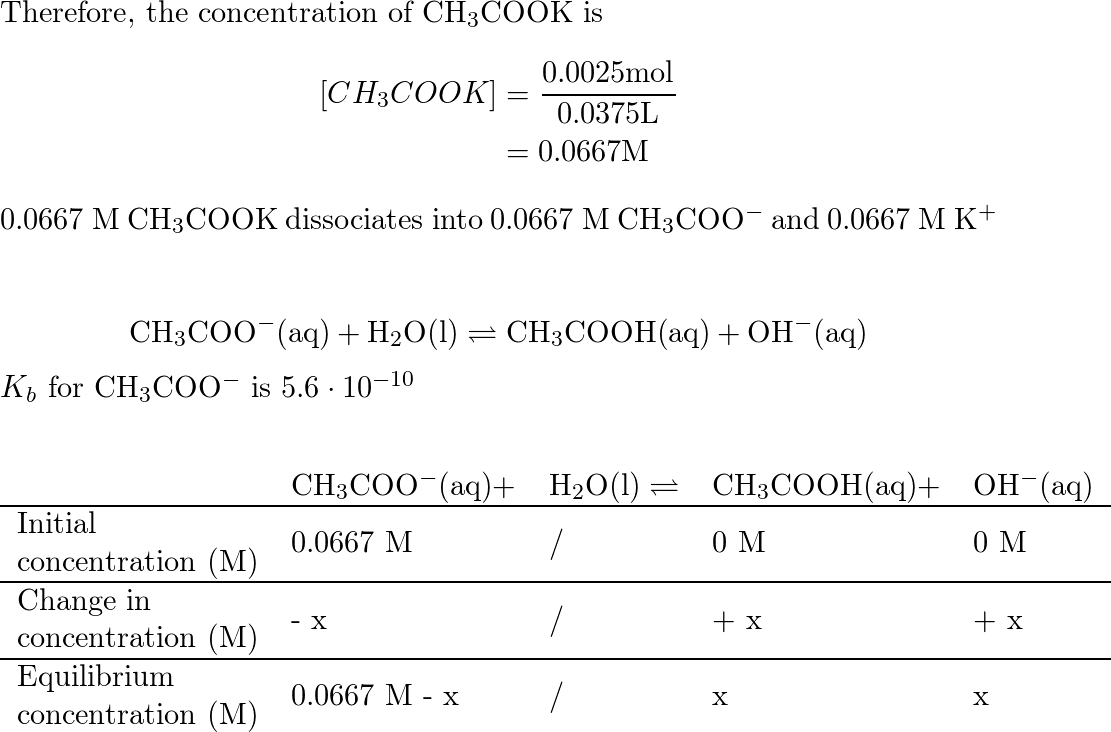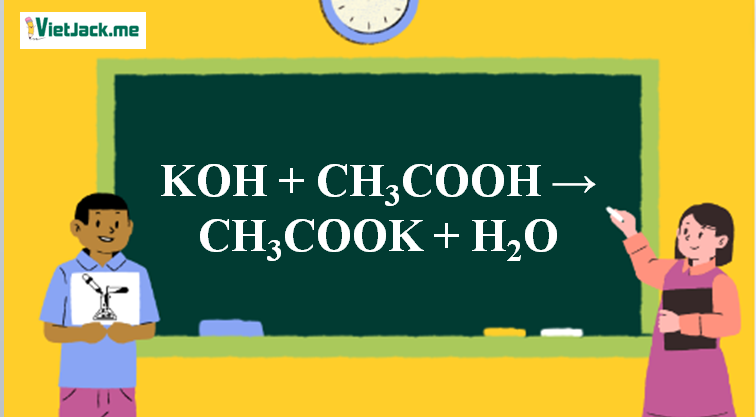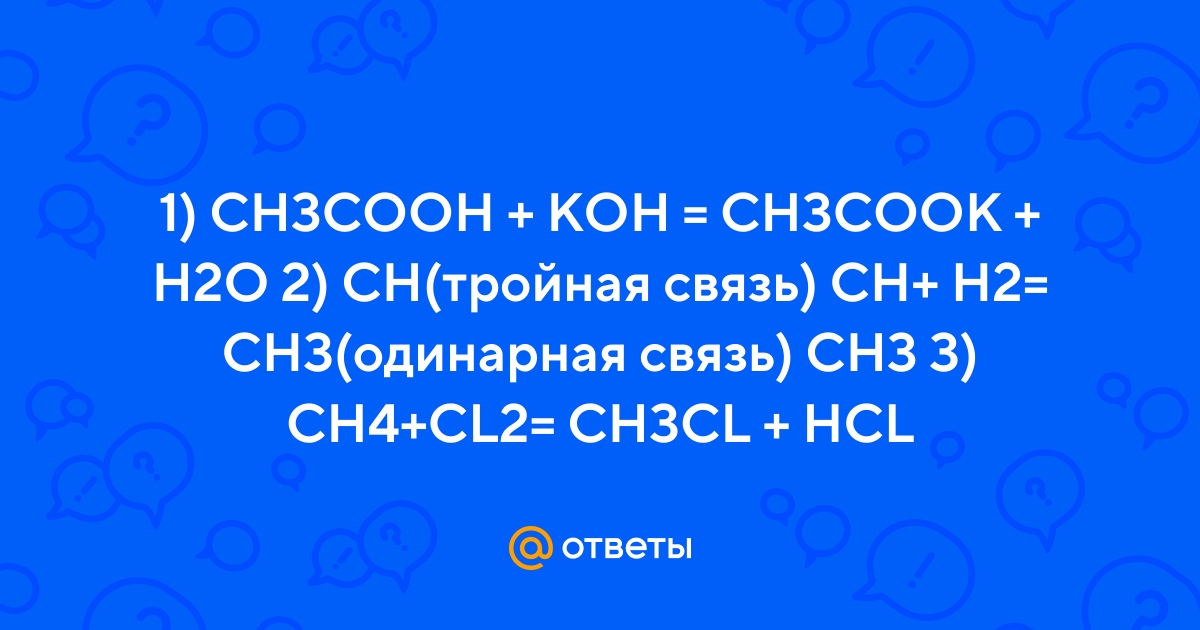
Ответы Mail.ru: 1) CH3COOH + KOH = CH3COOK + H2O 2) CH(тройная связь) CH+ H2= CH3(одинарная связь) CH3 3) CH4+CL2= CH3CL + HCL

Preparation of 3β-hydroxy-6-ethylchenocholane derivatives (subset B).:... | Download Scientific Diagram
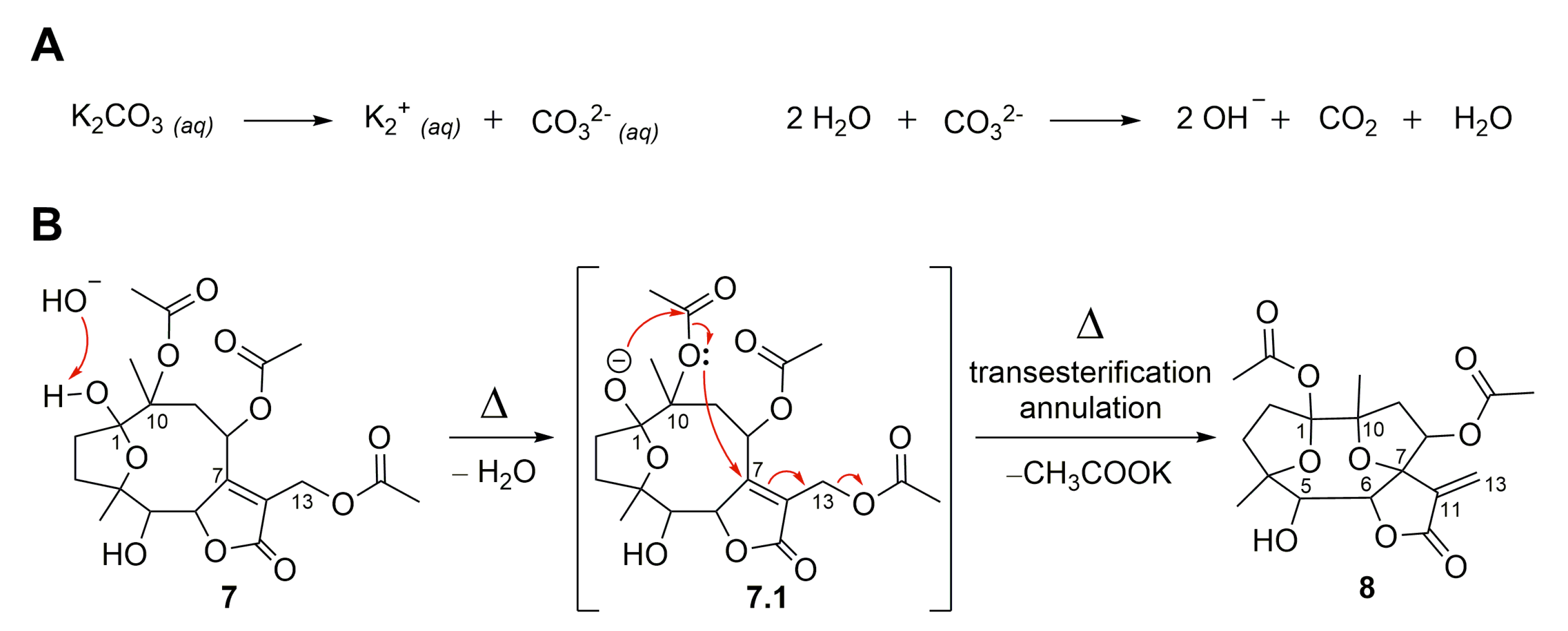
Molecules | Free Full-Text | Semisynthetic Sesquiterpene Lactones Generated by the Sensibility of Glaucolide B to Lewis and Brønsted–Lowry Acids and Bases: Cytotoxicity and Anti-Inflammatory Activities

SOLVED: QUESTION 4 POINT Identify the correct chemical reaction for the neutralization of ethanoic acid by potassium hydroxide. Select the correct answer below: CH3COOH + KOH â†' CH3COOK + H2O CH3COOH +