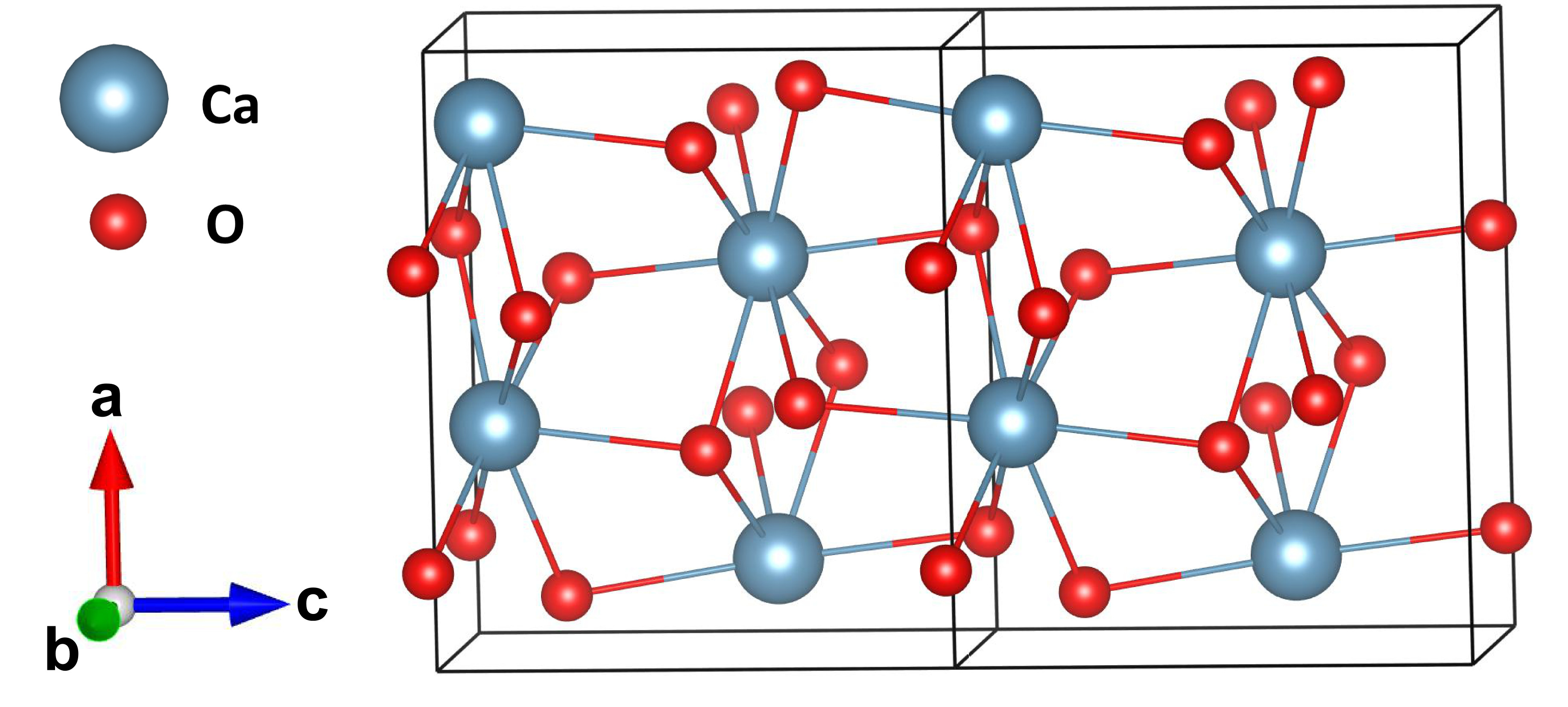
CaO2/UV Process for One-Step Phosphorus Removal and Recovery from Hypophosphite: Simultaneous Oxidation and Precipitation | ACS ES&T Water

When heated, metal hydroxides decompose to produce a metal oxide and water. Selected the correct balanced - brainly.com

Fabricating collagen films with oxygen-release capabilities: 1,7-octadiene PECVD encapsulation of calcium peroxide - American Chemical Society

C) Cal24 Pulice (d) All of these - Cl2 2 → A - Auto-oxidation Ca(OH)2 -H20 → CaCl2 + B Dry cao2 Identify B in the above reaction :
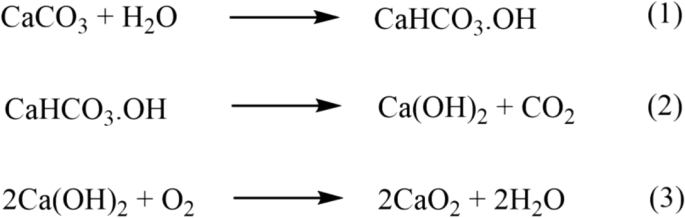
Novel iron sand-derived α-Fe2O3/CaO2 bifunctional catalyst for waste cooking oil-based biodiesel production | Environmental Science and Pollution Research

Influence of calcium peroxide on fermentation pattern and protozoa in the rumen: Archiv für Tierernaehrung: Vol 32, No 7-8
Consider the following reaction sequence:12.CaCO3(s)+ 2HCI(aq)CaCl2(aq) + CO2(g) + H20heatCaO(s) + H20(g).CaCO3(s)If the percentage yield of the 1st step is 80
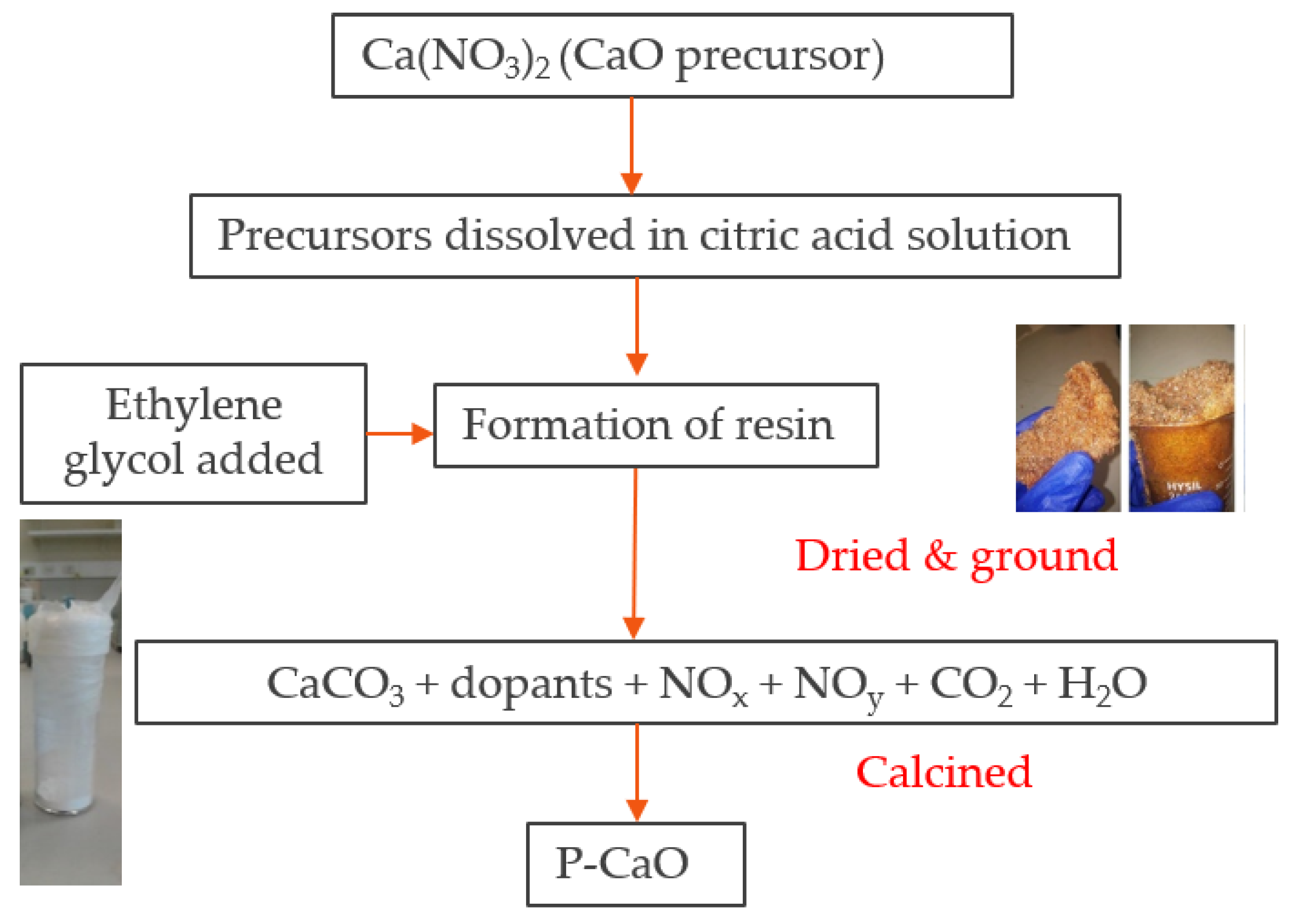
Applied Sciences | Free Full-Text | Comparative Kinetic Analysis of CaCO3/CaO Reaction System for Energy Storage and Carbon Capture
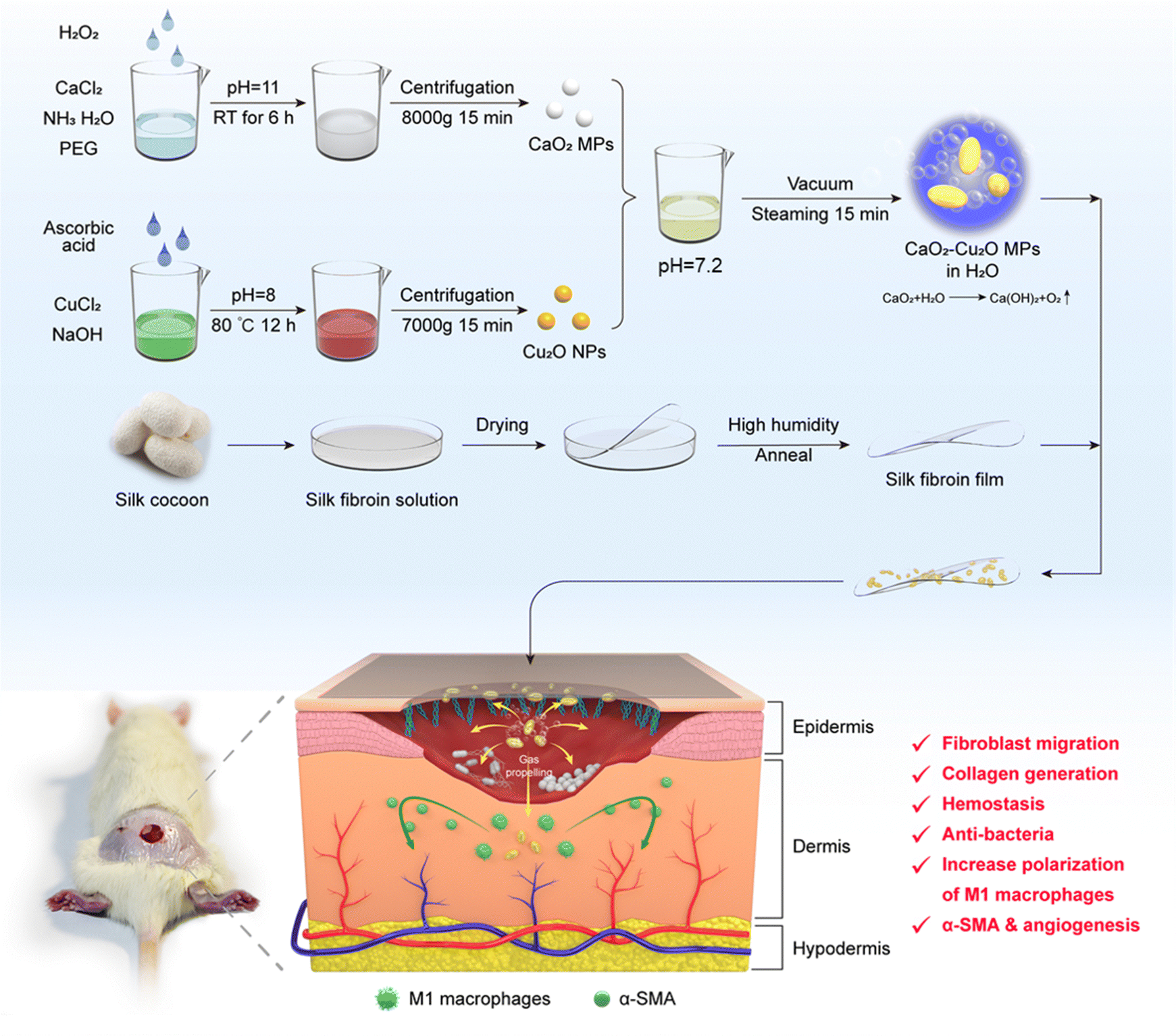
CaO 2 –Cu 2 O micromotors accelerate infected wound healing through antibacterial functions, hemostasis, improved cell migration, and inflammatory reg ... - Journal of Materials Chemistry B (RSC Publishing) DOI:10.1039/D3TB02335D

SOLVED: Given the data in the table below, calculate the enthalpy change (ΔH) for the reaction: Ca(OH)2 + 2 H3AsO4 â†' Ca(H2AsO4)2 + 2 H2O Substance ΔH (kJ/mol) Ca(OH)2 986.6 H3AsO4 900.4





![Punjabi] What type of reactions are represented by following equation Punjabi] What type of reactions are represented by following equation](https://static.doubtnut.com/ss/web-overlay-thumb/10335489.webp)