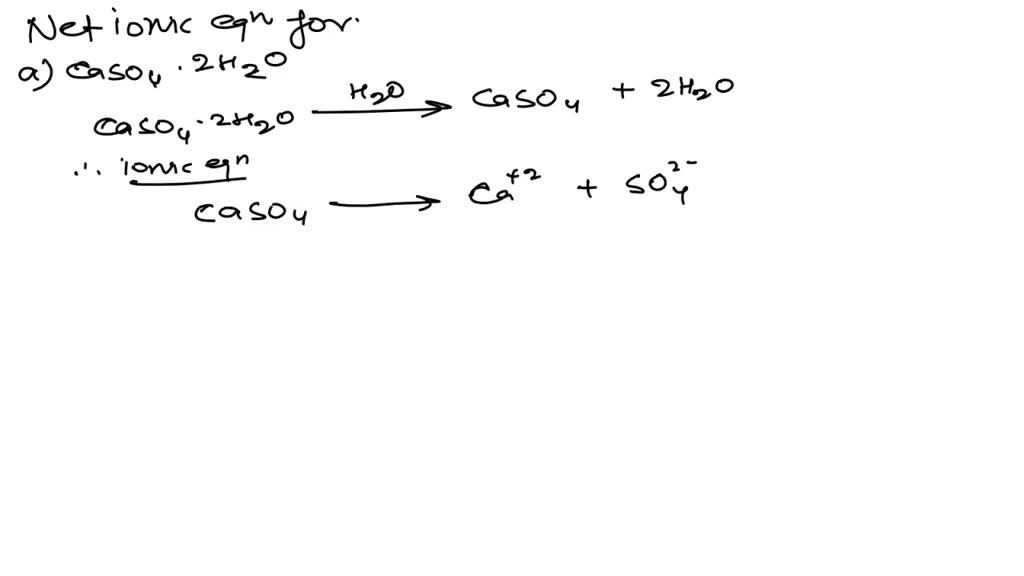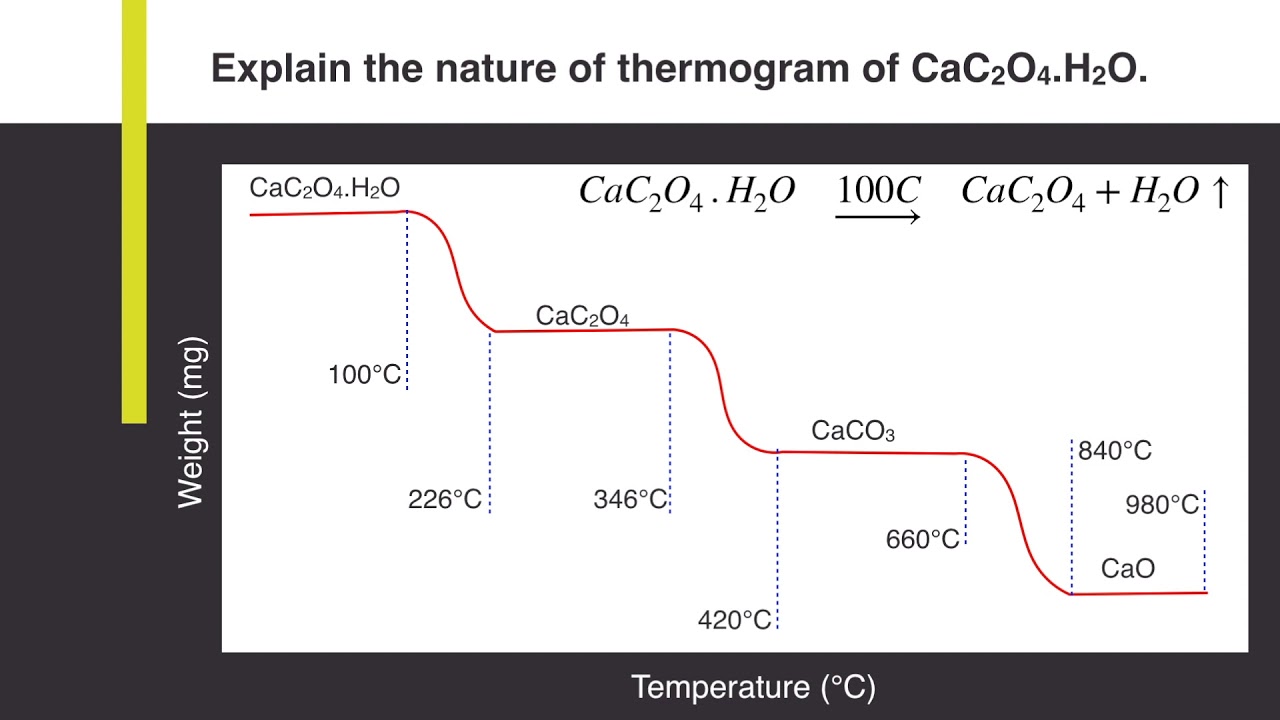
Explain the nature of thermogram of Calcium Oxalate Monohydrate (CaC2O4.H2O) | Analytical Chemistry - YouTube

SOLVED: Upon being heated, calcium oxalate monohydrate (CaC2O4 * H2O) undergoes a series of temperature-related changes, as shown in Equations 6-8. CaC2O4 * H2O(s) â†' CaC2O4(s) + H2O(g) (Eq: 6) CaC2O4(s) â†'
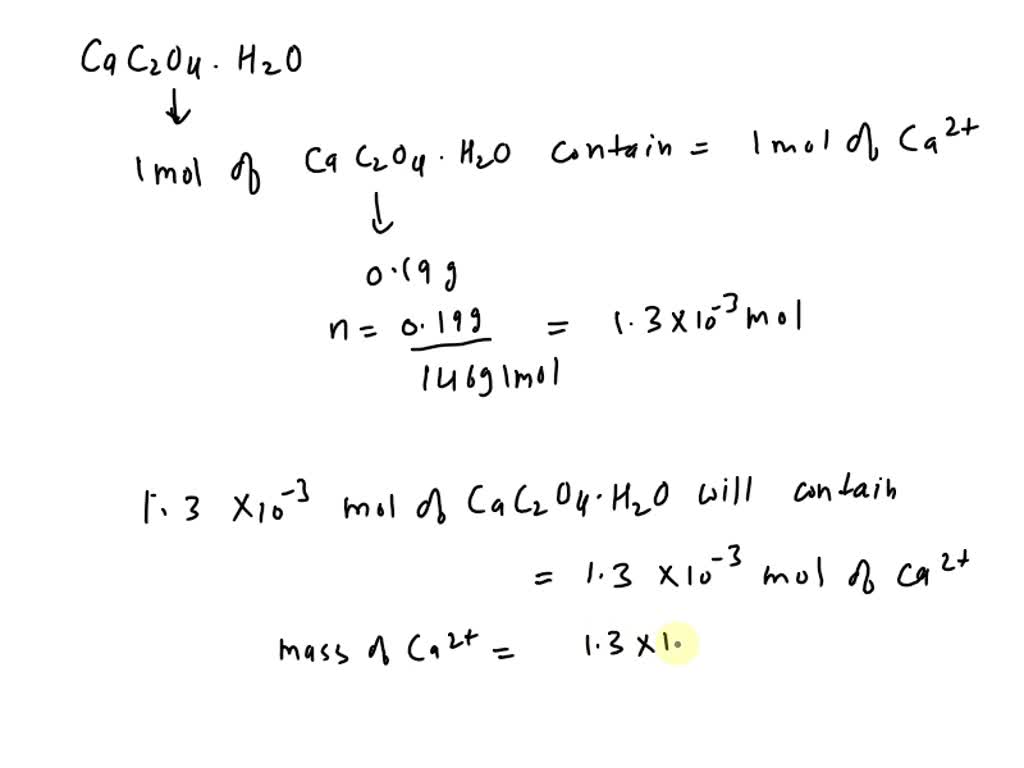
SOLVED: What is the concentration of calcium (Ca2+) in calcium oxalate monohydrate (CaC2O4·H2O)? Ca2+(aq) + C2O42-(aq) + H2O(l) â†' CaC2O4·H2O(s) The mass of calcium oxalate monohydrate is 0.190 grams. What is the [
Calcium oxalate, CaC2O4.H2O, is a sparingly soluble salt of analytical and physiological importance. - Sarthaks eConnect | Largest Online Education Community

Thermal Decomposition of CaC2O4·H2O Studied by Thermo-Raman Spectroscopy with TGA/DTA | Analytical Chemistry

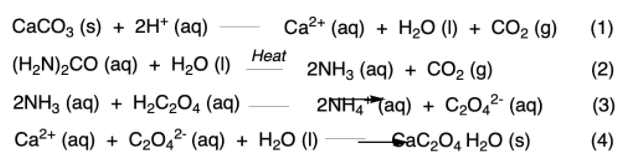
SOLVED: Gravimetric Determination of Calcium as CaC2O4 H2O Introduction Calcium ion can be analyzed by precipitation with oxalate basic solution to form CaC2O4 H2O. The precipitate is soluble in acidic solution because

Thermal Decomposition of CaC2O4·H2O Studied by Thermo-Raman Spectroscopy with TGA/DTA | Analytical Chemistry