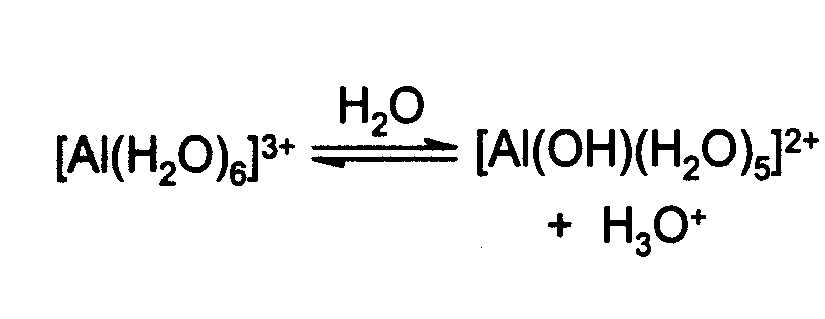SOLVED: For the reaction Al + H2O -> The products will be Al(OH)3, Al2O3 + H2O, and Al(OH)3. There is no reaction.
Al(H2O)6 3+ + H2O structures from MD simulations, all with d(Al-OW′)... | Download Scientific Diagram

Optical spectra obtained in Al-H2O reactor at HF plasma on and Argon... | Download Scientific Diagram
![Q.52 In the following [Al(H2O).)] + HCO3- [Al(H2O),OH)2 + HCO, (b) species behaving as Bronsted - Lowry acids are: Q.52 In the following [Al(H2O).)] + HCO3- [Al(H2O),OH)2 + HCO, (b) species behaving as Bronsted - Lowry acids are:](https://toppr-doubts-media.s3.amazonaws.com/images/2164712/a42abbba-24aa-49e2-9c74-16ac3879c732.jpg)
Q.52 In the following [Al(H2O).)] + HCO3- [Al(H2O),OH)2 + HCO, (b) species behaving as Bronsted - Lowry acids are:
![SOLVED: In the reaction Al3+ + 6H2O → [Al(H2O)6]3+, what does H2O act as? Bronsted-Lowry acid Lewis acid Bronsted-Lowry Base Lewis base SOLVED: In the reaction Al3+ + 6H2O → [Al(H2O)6]3+, what does H2O act as? Bronsted-Lowry acid Lewis acid Bronsted-Lowry Base Lewis base](https://cdn.numerade.com/ask_previews/29288c5c-bf0a-455e-8d4b-2c5553f62277_large.jpg)
SOLVED: In the reaction Al3+ + 6H2O → [Al(H2O)6]3+, what does H2O act as? Bronsted-Lowry acid Lewis acid Bronsted-Lowry Base Lewis base

SOLVED: The hydrated Al3+ ion, Al(H2O)6^3+, is a weak acid in water. What are the products of its reaction with H2O? Al(H2O)6^3+(aq) + H2O(l) â†' Al( H2O)5(OH)2^-(aq) + H3O+(aq)
 explains the formation of HO• radicals. | Semantic Scholar Figure 2 from Mechanism of Al(3+)-catalyzed oxidations of hydrocarbons: dramatic activation of H2O2 toward O-O homolysis in complex [Al(H2O)4(OOH)(H2O2)](2+) explains the formation of HO• radicals. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7caf468b460637902cf07e52ef61717b552aba94/5-Figure2-1.png)
Figure 2 from Mechanism of Al(3+)-catalyzed oxidations of hydrocarbons: dramatic activation of H2O2 toward O-O homolysis in complex [Al(H2O)4(OOH)(H2O2)](2+) explains the formation of HO• radicals. | Semantic Scholar
![SOLVED: In Al3+ + 6H2O â†' [Al(H2O)6]3+, what does H2O act as? Lewis base, Bronsted-Lowry base, Bronsted-Lowry acid, Lewis Acid 2. Correctedtext: What will happen if you remove a reactant from a SOLVED: In Al3+ + 6H2O â†' [Al(H2O)6]3+, what does H2O act as? Lewis base, Bronsted-Lowry base, Bronsted-Lowry acid, Lewis Acid 2. Correctedtext: What will happen if you remove a reactant from a](https://cdn.numerade.com/ask_previews/89fd73fd-55ff-4e1c-b8f2-49c556eb68c1_large.jpg)
SOLVED: In Al3+ + 6H2O â†' [Al(H2O)6]3+, what does H2O act as? Lewis base, Bronsted-Lowry base, Bronsted-Lowry acid, Lewis Acid 2. Correctedtext: What will happen if you remove a reactant from a
![OneClass: What is [Al(H2O)5(OH) 2+] in a 0.15 M solution of Al(NO3)3 that contains enough of the stro... OneClass: What is [Al(H2O)5(OH) 2+] in a 0.15 M solution of Al(NO3)3 that contains enough of the stro...](https://prealliance-textbook-qa.oneclass.com/qa_images/homework_help/question/qa_images/108/10827527.jpeg)